 The solenoid that generates the magnetic field has a diameter of 10 cm. Three experiments led him to this. The electrical discharge through the gases could be observed only at very low pressures and at very high voltages. A cathode ray tube is made of glass containing two thin pieces of metal (called electrodes) sealed in it. The simulation below uses metal plates 10 long with a spacing $d=$ 2 cm. He advanced the idea that cathode rays are really streams of very small pieces of atoms. Sub-atomic particles Discovery of Electron Electron was discovered by J J Thomson by Cathode ray discharge tube experiment. The force on an electron due to the electric field is $\vec. The electrons were accelerated in the $x$-direction, the electric field was in the $y$-direction and the magnetic field was in the $z$-direction. Thomson, a scientist, began working with cathode ray tubes in the late 1800s. Some of the electrons pass through a small hole in the plate and form and electron beam that travels to a region where an electric field and a magnetic field was present. The electron charge-to-mass ratio was measured by accelerating the electrons through a voltage $V_x$ towards a positively charged plate. He determined that the negatively charged particles (electrons) were much lighter than the positively charged particles. College, he applied and received a scholarship to Cambridge University in London. By balancing the effect of a magnetic field on a. 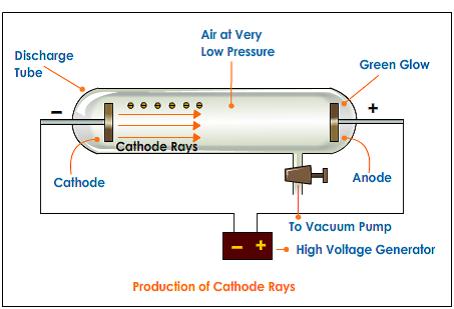
Thomson performed experiments to show that atoms consisted of sub atomic particles that had positive and negative charges. Thomson found that the cathode rays can be deflected by an electric field, as shown below. Thomson's experiment to determine the charge-to-mass ratio of electrons of many of energy scientists in an that Cathode Ray Experiment J.J. In his experiment, he placed two plates of opposite charges near the cathode rays. Thomson: Plum-pudding Model An atom is composed of negative electrons. Thomson began experimenting with cathode ray discharge tubes and studied the properties of the cathode ray. In 1913 Thomson published an influential monograph urging chemists to use the mass spectrograph in their analyses.J. In 1897, Joseph John Thomson, a British physicist proved that atoms are not the fundamental unit of matter. His nonmathematical atomic theory-unlike early quantum theory-could also be used to account for chemical bonding and molecular structure (see Gilbert Newton Lewis and Irving Langmuir). The apparatus of his experiment is called the cathode-ray tube (CRT). How many electrons does it have around the nucleus 30 The number of protons and electrons are equal in an uncharged atom. An uncharged zinc atom has 30 protons and 35 neutrons in its nucleus. Thomson determined that cathode ray particles were common to all matter. These particles later were named electrons. Thomson determined the mass-to-charge ratio of cathode ray particles. In 1897, he showed that cathode rays were composed of very small negatively charged particles. He was well-known for the discovery of the electron. Of all the physicists associated with determining the structure of the atom, Thomson remained most closely aligned to the chemical community. Sir Joseph John Thomson was a British physicist and Nobel Laureate. He was a good lecturer, encouraged his students, and devoted considerable attention to the wider problems of science teaching at university and secondary levels. Even though he was clumsy with his hands, he had a genius for designing apparatus and diagnosing its problems. 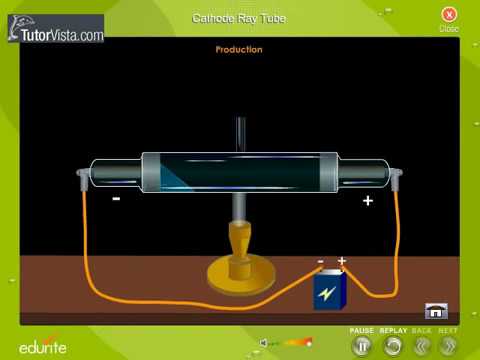
In 1884 he was named to the prestigious Cavendish Professorship of Experimental Physics at Cambridge, although he had personally done very little experimental work. This is one of the original vacuum tubes used by the Cambridge professor of physics Joseph John Thomson (1856-1940) to. Thomson repeated Hertz’s experiment with a better vacuum in 1897. He was then recommended to Trinity College, Cambridge, where he became a mathematical physicist. Instead young Thomson attended Owens College, Manchester, which had an excellent science faculty. His father intended him to be an engineer, which in those days required an apprenticeship, but his family could not raise the necessary fee. Ironically, Thomson-great scientist and physics mentor-became a physicist by default. From "The Growth of Physical Science," by Sir James Hopwood Jeans (Cambridge: Cambridge University Press, 1948) Early Life and Education 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
